Welcome to the Daggett Research Group.

Principle Investigator
Professor, UW Bioengineering
Founder and CEO, AltPep Corp.
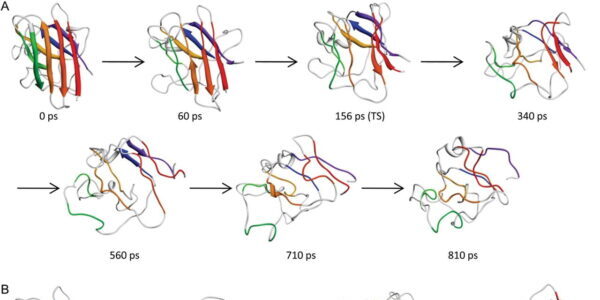
Our group is currently focused on protein unfolding and disease where we combine both computational and experimental approaches. We have developed a database to simulate representatives of all known protein folds, an effort we call Dynameomics, which has led to the largest collection of simulations and protein structures in the world.
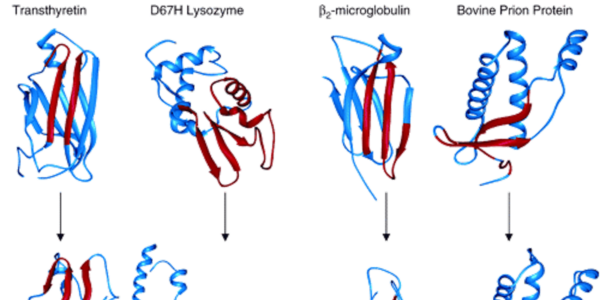
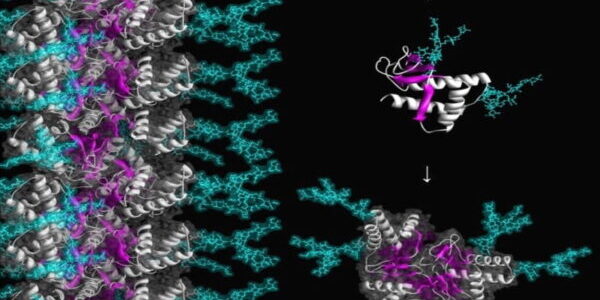
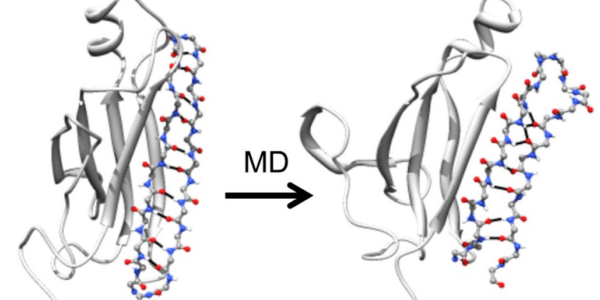
We also pursue simulations of protein unfolding related to amyloidogenesis. We found that amyloidogenic peptides and proteins under amyloidogenic conditions changed structure and formed a novel, nonstandard secondary structure, which we dubbed α-sheet. We proposed that α-sheet structure is linked to toxicity during amyloidogenesis, and have developed an α-sheet library to test our peptide inhibitor designs in 1) mammalian amyloid disease systems, and 2) bacteria that use amyloid fibrils for biofilm stability to evade the host immune response and antibiotics. Our compounds inhibit aggregation and toxicity by binding the toxic oligomers that form en route to mature amyloid.